The Thesis: The Challenge & The Goal
The late-stage development of a critical monoclonal antibody (mAb) required the immediate transfer of the entire upstream and downstream process from a pilot lab to a new, external Contract Development and Manufacturing Organization (CDMO).
The core challenge was an aggressive 9-month timeline to validated GMP batch production. The goal was 100% adherence to this timeline while navigating the regulatory complexity of a new site and ensuring zero critical process deviations.
My Role & Contribution
I served as the Process Lead and Project Management Integrator, bridging the gap between technical specifications and organizational execution.
- Role: Lead Process Development Engineer & Project Integrator
- Developed the integrated project schedule (WBS) and managed all critical path activities.
- Implemented a formal risk register (FMEA) focusing on Critical Process Parameters (CPPs).
- Negotiated across R&D, QA, and Operations to secure agreement on the Process Control Strategy.
- Led the final Process Performance Qualification (PPQ) campaign to 100% success.
Visual Evidence: Execution & Risk Control
Project Timeline & Risk Burn-Down
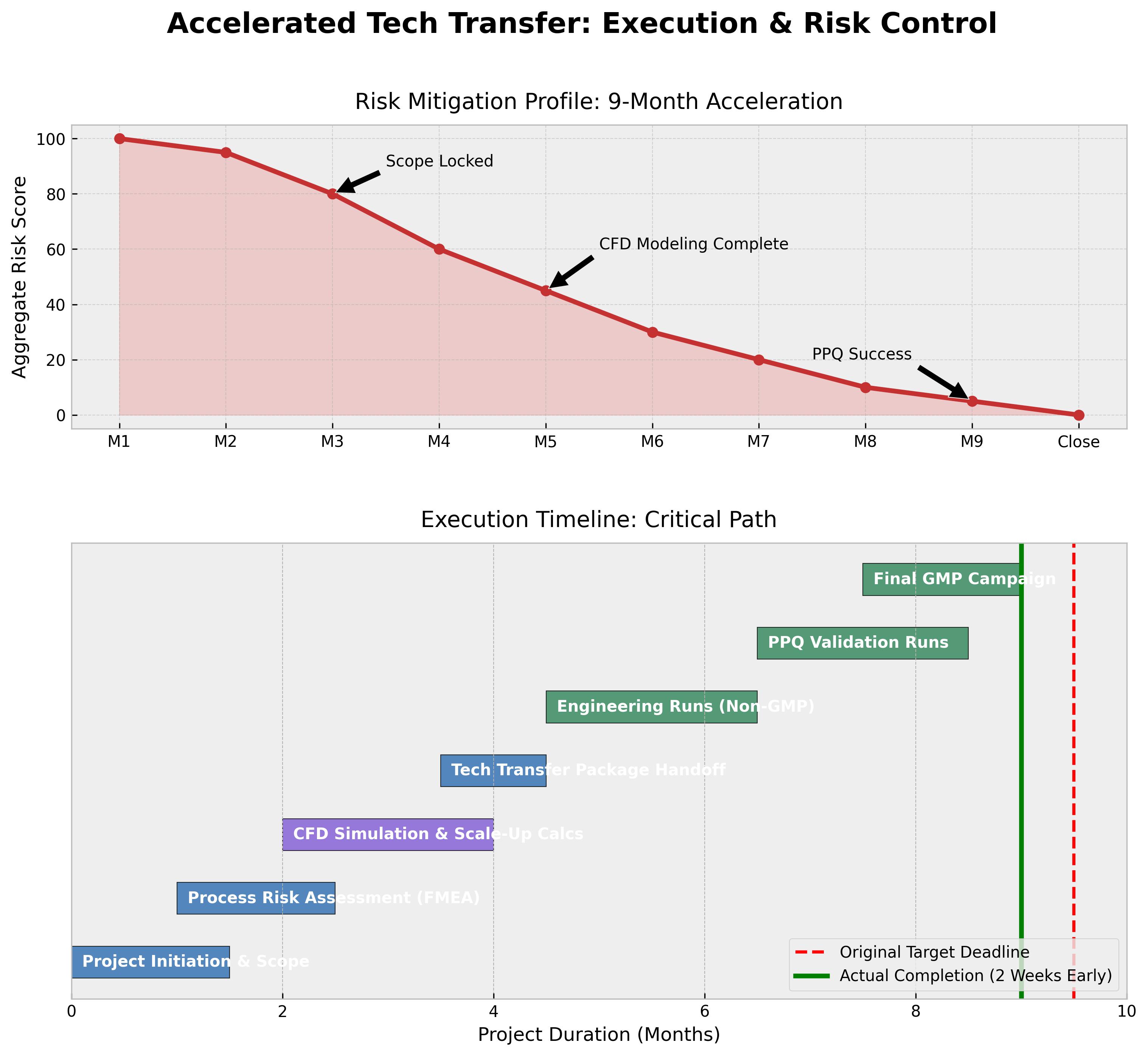
Figure 1: The dual approach of aggressive risk mitigation (top) enabled the team to maintain velocity on the critical path (bottom), resulting in early project completion.
The Impact: Results & Metrics
This project set a new company benchmark for external transfers:
- Timeline Success: Delivered the first three GMP batches 2 weeks ahead of the 9-month target.
- Cost Control: Executed the project 5% under budget through proactive resource leveling.
- Quality Assurance: Achieved 100% adherence to all Critical Process Parameters (CPPs) during validation.
- Organizational Influence: The communication framework established here became the new global SOP for tech transfers.
Methodology & Tools
- Project Management (WBS, Gantt, Risk Register)
- CFD Simulation (Mixing & Scale-up)
- JMP Pro (Statistical Analysis)
- DMAIC & Stage-Gate Frameworks